Hydrogen periodic table3/19/2023  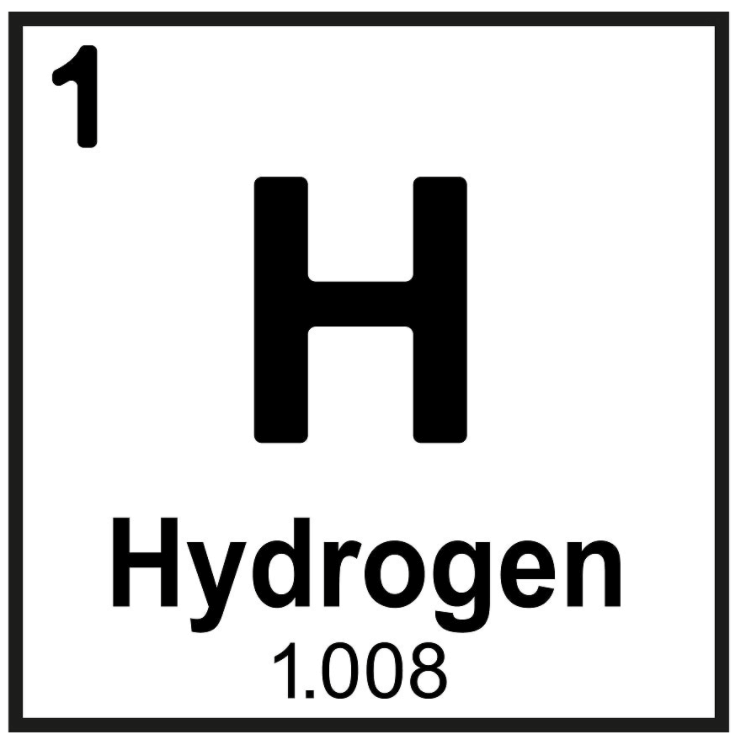
It is also placed with the alkali metals because it can lose its one e- to form H + simply such as the alkali metals. Hydrogen atoms contain the re-configuration 1s1, and it is the first element to be placed according to the rule. Hydrogen resembles the alkali metals in electronic configuration. In a few tables, it is placed with alkali metals (which is above Sodium), and in a few others, it is lonely placed at the top (Randomly, Just above the first Period). Generally, in the periodic table, Hydrogen does not have a fixed position. The Reason Behind Placing the Hydrogen Atom at First in the Periodic Table When the hydrogen atom loses electrons, the size of its nucleus decreases and almost becomes 1.5 × 10 -3 pm, which is much smaller when compared to the atomic sizes of the normal metals, and therefore the hydrogen ion does not freely exist in nature. Thus, in the periodic table, great thought has to be given for the hydrogen position. Though hydrogen atoms exhibit a lot of resemblance to both alkali metals and halogens, both are very different. It also exists as a diatomic molecule similar to that of halogens (for example, chlorine Cl 2 ) a single hydrogen bond exists when the H 2 molecule is formed. For example, ΔiH of lithium is given as 520 kJ mol -1, hydrogen is given as 1312 kJ mol -1, and for fluorine, it is given as 1680 kJ mol -1. By looking in terms of the ionization enthalpy, it is found that hydrogen resembles more halogens compared to alkali metals. 
Whereas unlike the alkali metals, it contains a very high ionization enthalpy, and hence it lacks metallic characteristics under regular conditions. Moving on to the formation of compounds, hydrogen produces oxides, sulphides, and halides resembling alkali metals. By taking a look at these properties, the position of hydrogen in the periodic table is the major question. When a hydrogen atom loses an electron and produces a cation, it resembles the alkali metals whereas, when it gains an electron and becomes a uni-negative ion, it represents similarity to the halogens. This character is mostly the same as that of the halogen family (ns 2, np 5 ), and is also short of one electron for the completion of the electron octet in their shells. Also, it can attain helium noble gas configuration by accepting an electron. The structure of hydrogen is similar to that of alkali metals (ns 1 ), which contains one electron in their outermost shell. With the wide number of properties, it is seen that Hydrogen is quite different from others while also showing a lot of similarities It is due to these similarities that are found that Hydrogen has a Position in The Periodic Table that is quite a different form above and is placed singly. Hydrogen has a lot of properties that are similar to a lot of the elements in the periodic table. 
This hydrogen element is widely used not only in industries but also in various daily life materials that are used. One of the smallest and the first element of the periodic table is hydrogen. The elements' placement in the periodic table is according to their electronic configuration. Therefore only 1 electron is available in its outermost shell. 
Hydrogen is defined as the first element of the periodic table because its atomic number is 1, which means it contains only one single electron in its atom. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
